Published on 13.04.2022
Today, only 50% of patients suffering from genetic diseases have a diagnosis. One of the Institut Imagine's ambitions is to raise this figure to 80% in the coming years and thus reduce the diagnostic wandering of patients and their families. This will require the identification of new genetic mutations. In a new publication published on 6 April 2022 in the American Journal of Human Genetics, the Developmental Brain Disorders Laboratory, headed by Vincent Cantagrel, has discovered a new gene responsible for a rare cerebellum disease [1].
This study was initiated following the follow-up of a first patient, who died at the age of 25 months, with severely reduced muscle tone and an autonomic nervous system defect with breathing and swallowing disorders. MRI scans revealed a reduction in the size of his cerebellum, a subcortical brain structure at the back of the brain that is essential for motor functions such as learning to walk and balance, but also for higher-level cognitive processes such as attention and language. The developmental abnormality also affected the brain stem, the part of the brain that connects to the spinal cord.
No diagnosis could be made at that time. "Using a panel of already known genes, the doctors were unable to identify any genetic mutations that could explain this disease," summarises Marion Coolen, the publication's principal investigator. This prompted the researchers to carry out exome sequencing, an examination consisting of reviewing all the coding parts of the patient's DNA. "We found the same mutation in both copies of the PRDM13 gene," she explains. Thanks to collaboration with the Centre de référence des malformations et maladies congénitales du cervelet, the Bordeaux University Hospital, as well as with foreign laboratories, new patients carrying mutations in the PRDM13 gene were subsequently identified, with similar clinical manifestations.
A key role in balancing neuronal composition
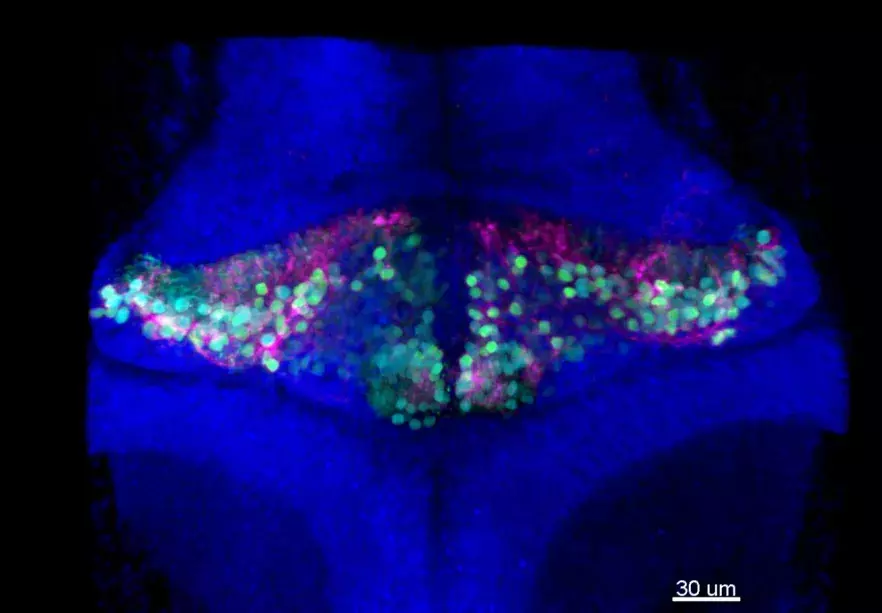
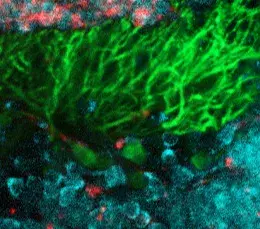
Although suspicious, this correlation did not allow us to affirm the direct involvement of the gene in the disease. Indeed, no role was known for PRDM13 in the embryonic development of the cerebellum and brainstem. By combining two techniques (in situ hybridisation and single cell transcriptomic analysis), the researchers were then able to show that PRDM13 is only expressed at very early stages of development, in a specific region of the cerebellum where the so-called "inhibitory" neurons are formed, in particular the Purkinje cells, which are very recognisable by their abundant branches (see photo). The PRDM13 gene thus seems to play a central role in the differentiation of immature cerebellar cells into inhibitory neurons, and thus in the balance of the neuronal composition of the cerebellum.
Our discovery has already been of immediate interest to families
In order to clarify the physiopathological mechanisms, the researchers used another model: the zebrafish. "Its cerebellum is very similar to ours both in its structure and in its cellular composition. Moreover, thanks to the analysis of Single Cell data carried out in collaboration with the Bioinformatics platform and Antonio Rausell's laboratory [Clinical Bioinformatics lab], we were able to highlight great similarities in the expression of PRDM13 in humans and zebrafish during development. This makes it a good model," explains Marion Coolen.
How zebrafish model helps to better understand the disease
The researchers then studied a line of fish with the same mutations as the patient and were able to observe a significant reduction in the number of Purkinje cells in the cerebellum, exactly as observed in the patient. The fish larvae also showed similar abnormalities in the brainstem. All of this suggests that the mutation in PRDM13 is responsible for the disease.
"Our discovery has already been of immediate interest to families," says the researcher. One family has already benefited from a prenatal diagnosis at Necker, which has led to the arrival of a healthy child, and other families are asking for it.
This work has also highlighted the essential role of PRDM13 in the development - as yet unknown - of structures involved in the regulation of cerebellum and autonomic nervous system functions. These structures could be involved in other pathologies such as sudden infant death syndrome. Finally, this work could also shed new light on the genetic programs that underlie the specification of neuronal types and how these programs readapt in the presence of a mutation such as that of PRDM13 patients.
The laboratory is currently working on the development of an organoid model, a kind of miniature cerebellum reproduced in vitro from patient stem cells, which would be ideal for further understanding these mechanisms. This could help identify new genes and key regulatory sequences that are potentially mutated in patients. In the long term, a better understanding of neurogenesis in the cerebellum could be exploited for therapeutic purposes.